ESSENTIAL OIL
STEAM DISTILLATION KITS
The BEST essential oil steam distillers

................. Click this image for info
Boiling Points
Everyone has boiled water at some point so everyone has a basic understanding of what is meant by boiling. We put a pot on the stove, go away for ten minutes, and come back to find the water boiling vigorously, ready for the pasta, eggs, or oatmeal to be put in.
To really watch what happens with water we must have a thermometer and some patience. When we put cool tap water into a beaker and place this on the stove and turn on the heat we may see ripples in the water as the water heats. These ripples are called Schleeren patterns and arise from light being diffracted off of the regions of water with different temperatures and hence different densities. This is exactly the same optical effect that gives rise to mirages. Since the water is being heated from below, the lighter, warm water will rise in the beaker causing cooler surface water to flow to the bottom.
At some temperature, usually around 80 oC, small bubbles will be observed on the inner surface of the beaker. Many people confuse this with boiling. These bubbles are the air that was dissolved in the water at room temperature coming out of solution. Air becomes less soluble in water as the temperature goes up.
If we continue to heat the water we will begin to observe bubbles form and collapse on the bottom of the beaker. These are the precursors to true boiling. The water on the bottom surface of the beaker is heated to the point where it converts to vapor, but this vapor cools rapidly as it expands and as it encounters slightly cooler water just above the bottom of the beaker. As the bulk water in the beaker becomes hot the bubbles begin to break free of the bottom and rise. Only when the water in the beaker is uniformly hot will the bubbles rise from the bottom and break free at the top. This is often called a rolling boil and is the point of true boiling. The phenomenological definition of boiling is the existance of sustained bubbles that break free of the surface.
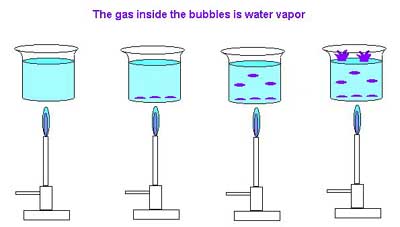
Let's look more closely at this process. The bubbles that form at the boiling point contain water vapor. In order for a bubble to exist the pressure of the vapor in the bubble must be pushing against the water with exactly the same force that the water is pushing back. Assuming that we have a normal beaker and just a few cm of water, almost all of the pressure being exerted by the water is air pressure. Hence for the bubble to exist the vapor pressure must be equal to atmospheric pressure. We define a "normal" boiling point as the boiling point of the liquid at 1 atm, or another way of saying this is that the "normal" boiling point is the temperature at which the liquid's vapor pressure is equal to 1 atm.
In other studies it has been established that vapor pressure is determined by both temperature and by intermolecular forces, and that the "normal" boiling point will reflect the intermolecular forces of the liquid.
Does "Boiling" Mean "Hot"?
If boiling occurs when the vapor pressure inside the bubble equals atmospheric pressure, what would happen if the atmospheric pressure would drop? The simple example of this is the case of a camper who is on the top of a 10,000 ft mountain. While water boils at 100 degrees C at sea level (where the atmospheric pressure is 760mm Hg) the atmospheric pressure at 10,000 ft altitude is about 530 mm Hg. Because the boiling point of water is about 90 degrees C at this altitude., not only will our camper find that his food will cook a little slower than normal, any attempt to boil water to kill germs and pathogens may be unsuccessful since the water isn't getting hot enough.
What about boiling things at increased pressures?
Pressure cookers take advantage of just this; they are equipped with a valve that lets gas escape only when the pressure inside the pot exceeds some fixed value. This valve is often set at 15 psi, which means that the water vapor inside the pot must reach a pressure of 2 atmospheres (1520mm Hg) before it can escape. Because water doesn't reach a vapor pressure of 2 atm until the temperature is 120 degrees C, it boils in this container at 120 degrees C, so things cook faster because they are hotter!
Boiling Chips, what are they and why do we use them?
Liquids often boil in an uneven fashion, or "bump" as it's called in chemistry. Bumping occurs frequently when you are distilling solvents or mixtures and there aren't any scratches or irregularities on the walls of the boiling pot that would help boiling bubbles to form. If this happens, a thing called superheating occurs- because the walls of
your boiling pot are smooth and bubbles of vaporized liquid cannot easily form, the temperature
of the liquid can actually rise above it's boiling point without boiling- the solution is now a superheated liquid. This can be dangerous because bubbles eventually will form, and when finally do, they usually erupt violently because so much of the liquid is just itching to boil but hasn't been able to, that it all goes bloooop and releases all of it's superheated boiling energy at once!
The result is that the entire solution turns into a mass of gas and bubbles, and with a resounding "thump" it can send a shockwave of undistilled boiling solution shooting up and through the entire distillation system- at the very least contaminating everything you've just distilled, and at worst it can actually cause the glassware to come apart or even break due to the sudden pressure wave!!
An example of this happening at home- there are many reports of people heating coffee in a microwave- and when the oven goes "ding" and the door is opened, there sits the nice cup of coffee... until they touch it and it unexpectedly goes "SPLAT"- half the cup of scalding coffee is suddenly all over the place inside the oven, on their hand and on the floor in a big scalding mess- what happened? the coffee was too still to boil and it just sat there superheated until they moved it, and at that point it bumped!!
Bumping is easily prevented by adding a few things called "boiling chips" to the liquid. Boiling chips are small, insoluble, stones (the size of grains of rice or sand) made of calcium carbonate, silicon carbide, or carbon (crushed coal) just to name a few. These stones contain many microscopic pores and/or or sharp points which provide a rough surface upon which boiling bubbles can form. When boiling chips are used, essentially all of the bubbles that rise through the solution will have formed on the surface of these chips.
A frequent exception to this rule is when boiling water- and for our water-based steam distllers we've found that boiling chips are not necessary
Never add boiling chips to a hot liquid, because it can cause immediate boiling over of the solution. If you forget to add boiling chips before you begin heating a solution, you must cool the solution before adding one to prevent product loss. "Porous" boiling chips cannot be re-used since the pores inside these stones become filled with liquid on cooling. "Sharp" boiling chips like silicon carbide or coal can often be reused until they become coated with guk and become innefective.
SO what does all of this have to do with herbs?
Tinctures are made with solvents, and some herbs and fragrent flowers are better extracted with solvents than by steam distillation for various reasons, (ie being destroyed by steam temperature). Often times, you'll want to remove some or all of the solvent from the extracted plant material, or concentrate your tincture that you've just made, and boiling it off is a good way to do it, as long as the boiling temperature of the solvent won't hurt your product. This is at least one procedure in which you'd want to use boiling chips.
Much of the above information was gleaned from, amongst others, http://www.chem.uidaho.edu/~honors/
Boiling and separating solvents vs system pressure, and azetropes
Vapor temperatures of solvents and liquids are published assuming they are at sea level. For example, if it's ethanol (alcohol) that you're boiling off, and you're at sea level, the vapors will be at 78 degrees C..... if it's water you're boiling off, the vapors will be at 100 degrees C
When you are distilling (boiling) mixtures containing solvents, you can tell what solvent is currently distilling in your setup by watching a thermometer that is placed in the vapor of the boiling mixture, and calculating what solvent is currently boiling away using the measured vapor temperature vs system pressure.
Now I chose these two solvents (yup, water is a solvent, and a great one at that!) because in the case where they are mixed, a funny thing happens... water and ethanol in particular form something called an azetropic mixture (also known as a constant-boiling mixture) which in simple terms means that the alcohol cannot be separated from the water completely...at somewhere around 95% pure, the mere act of boiling won't remove any more water. That's why you find "Everclear" at a maximum purity of 190 proof, or 95%, because it's impossible to remove any more of the water from it using distillation!!
So when you boil off a mixture of alcohol and water, expect temperatures somewhat different than the individual components alone...ie alcohol mixed with water can boil anywhere between 78.2-99.9 degrees C, depending on the ratio of alcohol and water.
Practical Example:
When you are separating a mixture of, say acetone and water, what you will experience is that the vapor temperature at the stillhead™ will go immediately to 56 degrees c, the vapor temperature of boiling acetone- and it will stay there until the acetone has all boiled away and there' s none of it left in solution. At this point the distillation will slow or even stop for a moment as the boiling solution quickly raises to the boiling point of water, 100c. Then the vapor temperature at the stillhead™ will stay right at 100c until the water's all gone. If you had a third component to the solution that had a higher boiling point than water, this would repeat and it would go to that tenperature.
When you're attempting to separate say, a 50-50 mixture of ethanol and water, what you'll find is that the initial boiling point is going to be somewhere closer to that of pure ethanol. But instead of sticking there througout the time period that you're boiling the ethanol off, you will find that the vapor temperature smoothly and constantly rises up and up until it's 100 degrees c.
What's happening is that the "starting" temperature is determined by the RATIO of alcohol to water in the boiling pot, and hence the ratio of ethanol to water that is evaporating- and as the alcohol goes away the ratio changes, thus the vapor temperature constantly rises, as does the amount of water that's being distilled along with the ethanol.
If you're distilling to separate ethanol, the purity of your distillate will be really high in the beginning and slowly become more and more dilute as the distillation progresses, because as ethanol leaves the boiling pot, so does water, in a ratio that gets higher and higher, perfectly alligned with the vapor temperature at any given moment. The less ethenol there is in the boiling pot, the higher the temperature of boiling is, and higher is the ratio of water to ethanol that is 'coming over".
So to obtain the highest purity of ethanol, you would want to decide a cutoff point and stop the distillation much earlier than you would normally think. For high purity you might want to cut off at 90 degrees. You won't get all the ethanol out of your vodka that way, but what you do obtain will be a lot less dilute than if you let it go to say, 95 degrees.
ALTITUDE
When you are boiling things at higher altitudes than sea level, the atmospheric pressure in the system is less than if you were at sea level. According to Boyle's law , the actual boiling temperature (and vapor pressure) of liquids will be less than they are at sea level!
There are many charts and nomographs published with which you can find the corrected boiling temperatures of solvents if you are distilling at higher altitudes or in vacuum distillation systems
The bottom line here is: if you are doing an otherwise normal atmospheric distillation and your thermometer just WON'T go as high as you think it ought to, take a moment and remember that if you live in the mountains, you shouldn't toss your thermometer in the trash, because it's doing exactly what it ought to!!
Here are some common solvent boiling points:
(Consider them all toxic but the ones labelled "toxic" are REALLY nasty...)
ACETONE (dimethyl ketone, ketone propanone, propanone)
bp 56.48 C
BENZENE (C6H6, benzol, phenyl hydride, coal naptha) (included for comparison and cautions...you wouldn't want it around yourself anyway)
bp 80.1 C,
TOXIC: 3000 ppm vapor considered high concentration, toxic via inhalation or skin absorption as well as oral ingestion, prolonged inhalation of low concentrations also toxic
NOTE: do not confuse with benzine which is a petroleum distillate
Caution: TOXIC: Benzene is a recognized leukemogen (causes leukemia)!
BUTANE (C4H10, n-butane, methylethyl methane, butyl hydride)
bp -0.5 C
CHLOROFORM (CHCL3, trichloromethane)
bp 62.26 C,
DICHLOROMETHANE see methylene chloride
DECANE (CH3(CH2)8CH3)
bp aprox 174 C
DIETHYL ETHER (C2H5-O-C2H5, ether, ethyl ether, anesthesia ether, ethyl oxide)
bp 35 C
ETHYL ALCOHOL (ethanol, methyl corbinol, spirit of wine, grain alcohol, Everclear, 95%)
bp 78.32 C,
note: there have been reports of people using denatured alcohol as a solvent.
NOT a good idea if the result is for human or animal use.
May be ok if it is denatured only with something that will be eliminated when the solvent is boiled off (e.g. with methyl alcohol).
If you don't ABSOUTELY know that you can eliminate the denaturant, then don't use denatured alcohol!
Results varying from death to blindness could result!
KEROSENE
bp 175-325
HEPTANE (C7H16)
bp aprox 98 C
HEXANE (C6H14)
bp aprox 69 C
ISOPROPYL ALCOHOL (dimethyl alcohol, sec-propyl alcohol, isopropanol)
bp 80.3 C,
METHYL ALCOHOL (methanol, wood alcohol)
bp 64.8 C,
TOXIC !! death to blindness can result from it's ingestion!
METHYLENE CHLORIDE (CH2Cl2, dichloromethane)
bp 39.8
TOXIC!!
NAPTHA, V.M. & P. (benzine, 76 degree naptha)
bp 100-140 C,
NAPTHA, V.M. & P.
bp 138-165 C,
NONANE (C9H20, n-nonane)
bp aprox 151 C
OCTANE (C8H18)
bp aprox 126 C
PENTANE (C5H12, n-pentane)
bp aprox 36 C
PETROLEUM SPIRITS (petroleum benzine, petroleum naptha, light ligroin, petroleum ether, mineral spirits)
bp 35-180 C,
alpha-TRICHLOROETHANE (CH3CCl3, 1,1,1-trichloroethane, methyl chloroform)
bp 74.1 C,
TOLUENE (C6H5CH3, methylbenzene, phenylmethane, toluol)
bp 110.4 C
TURPENTINE (spirit of turpentine, turpentine gum, turpentine oil)
bp 154-170 C
WATER (H2O)
bp 100 C (212 F)
Amazing solvent that you can actually drink!
XYLENE (C6H4(CH3)2)
(m-xylene) bp 139 C
(o-xylene) bp 144.4 C
(p-xylene) bp 138.3 C
Ā Ā